Phase 1 Clinical Trial Protocol Template
Clinical trial protocol documents template division of aids daids for daids protocol development guidance see the clinical trial protocol documents manual v1 0 this document is a daids sample protocol template which is the preferred daids protocol format.
Phase 1 clinical trial protocol template. International conference on harmonisation ich guidance for. For phase 1 trials. Generic protocol documents and instructions for ctep studies instructions for submitting protocol documents to ctep pdf step by step guide for submitting esubmission ready documents to ctep pdf generic protocol template ms word updated february 5 2020. Early developmental protocols should specify in detail all the elements of the. The nih also released a secure web based e protocol writing tool that allows investigators to generate a new protocol using the nih fda phase 2 and 3 ind ide clinical trial protocol template.
The goal of this template is to assist investigators to write a comprehensive clinical trial protocol that meets the standard outlined in the. The template follows the international conference on harmonisation ich e6 r2 good clinical practice and is available as a word document. Suggested templates for phase 1 and 2 clinical trials. The first type of trials are phase 2 and 3 clinical trial protocols that require a food and drug administration fda investigational new drug ind or investigational device exemption ide application. Mercuric1 confidential general contact information trial office octo mercuric trial office oncology clinical trials office octo department of oncology the university of oxford oxford cancer and haematology centre.
Phase 1 clinical trial protocol template for nonclinical research or clinical trials that are phase 0 or phase 1 use this free template. This template may also be useful to others developing phase 2 and 3 ind ide clinical trials. Phase 1 or nonclinical trials do not require the same amount of detail as a full study protocol. Takeda development centre europe ltd. Protocol amendment a phase 1 randomized double blind placebo controlled safety tolerability and pharmacokinetic study of escalating single and multiple doses of tak 653 in healthy subjects phase 1 tak 653 escalating single and multiple dose study in healthy subjects sponsor.
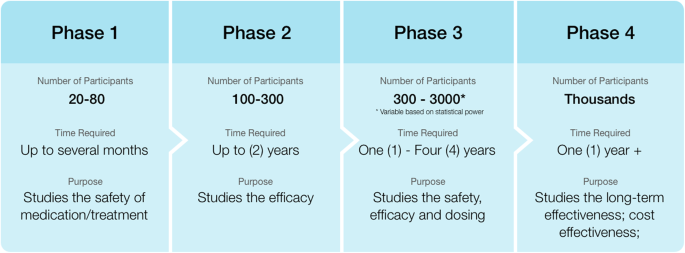
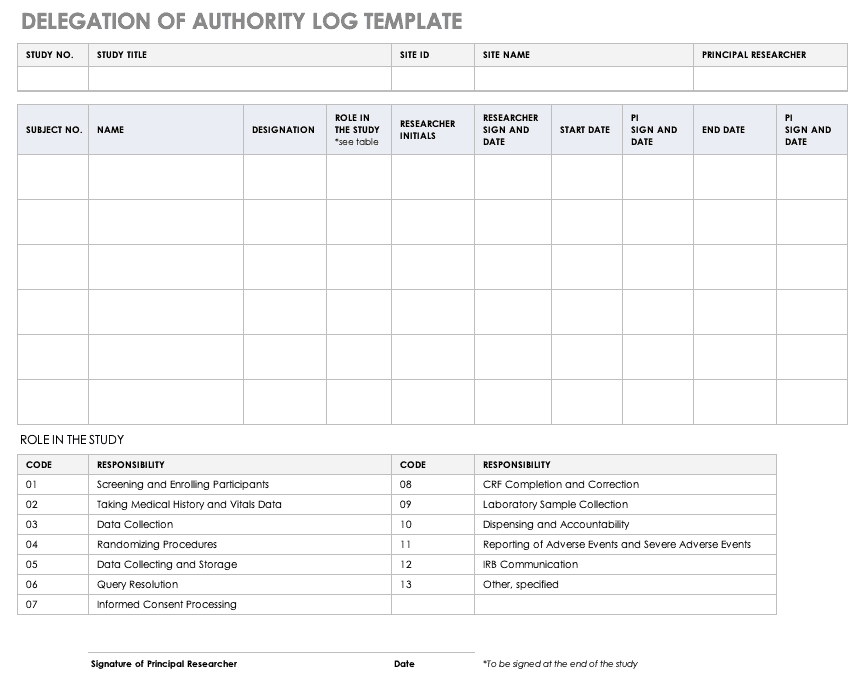
If the drug successfully passes through phases i ii and iii it will usually be approved by the national regulatory authority for use in the general population. This policy does not apply to informed consent ic development or daids ic templates. Phase iv trials are post marketing or surveillance. Mercuric1 protocol v8 0 11sep2017 protocol template v3 0 18feb2013 page 1 of 112. This protocol template aims to facilitate the development of two types of clinical trials involving human participants.
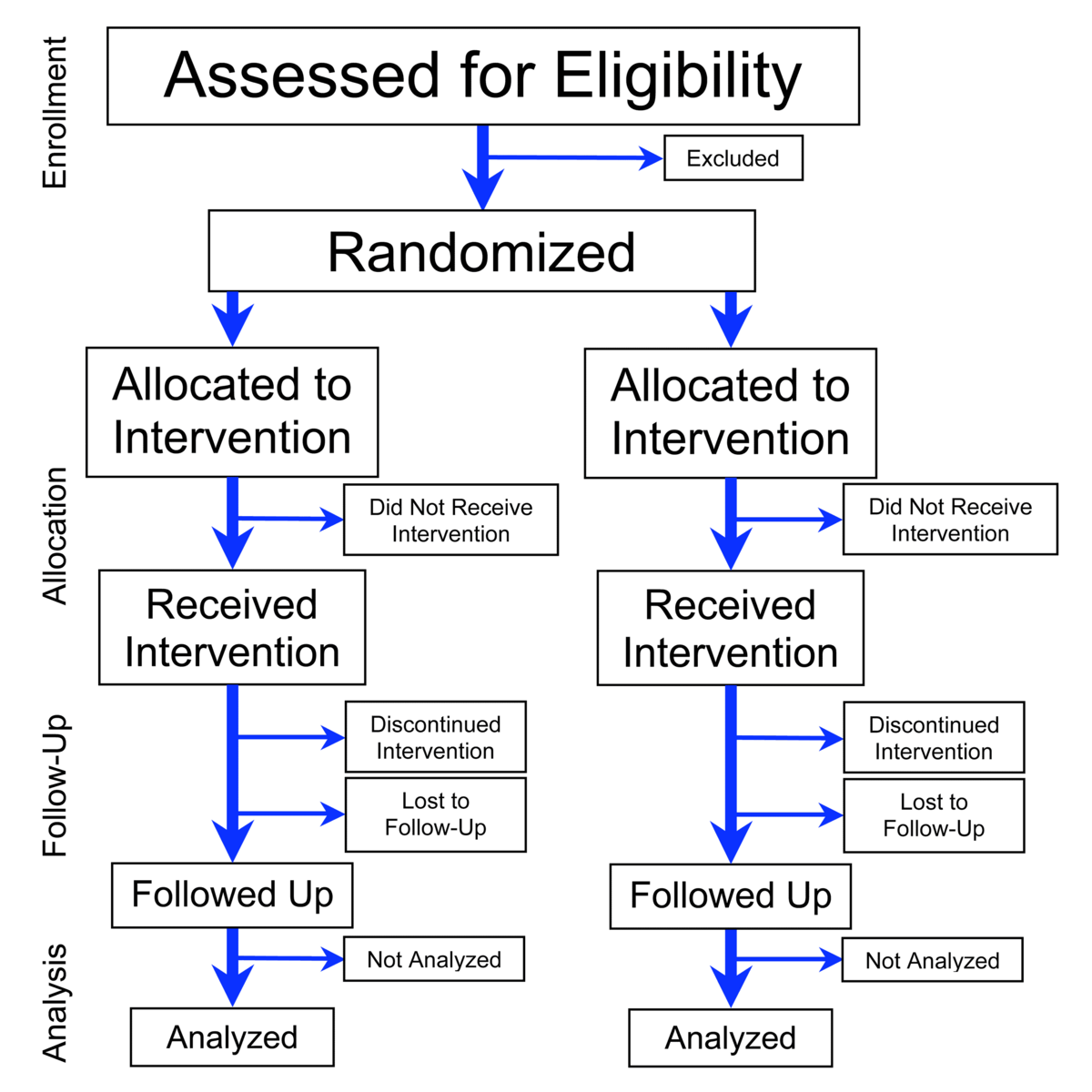
The drug development process will normally proceed through all four phases over many years.
eu mdr declaration of conformity template email template for confirming you have completed a task environment of care risk assessment template environmental health and safety management system template do you have to use a template on squarespace employment application template for truck driver employee pto tracker excel template format